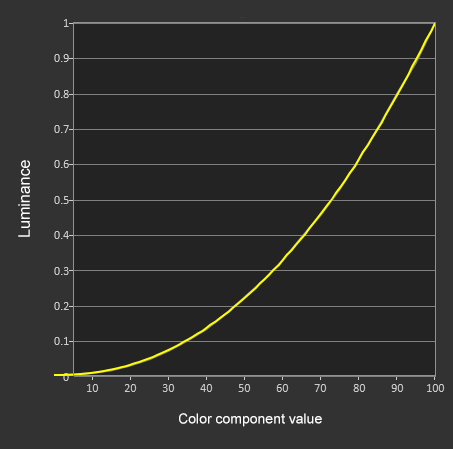
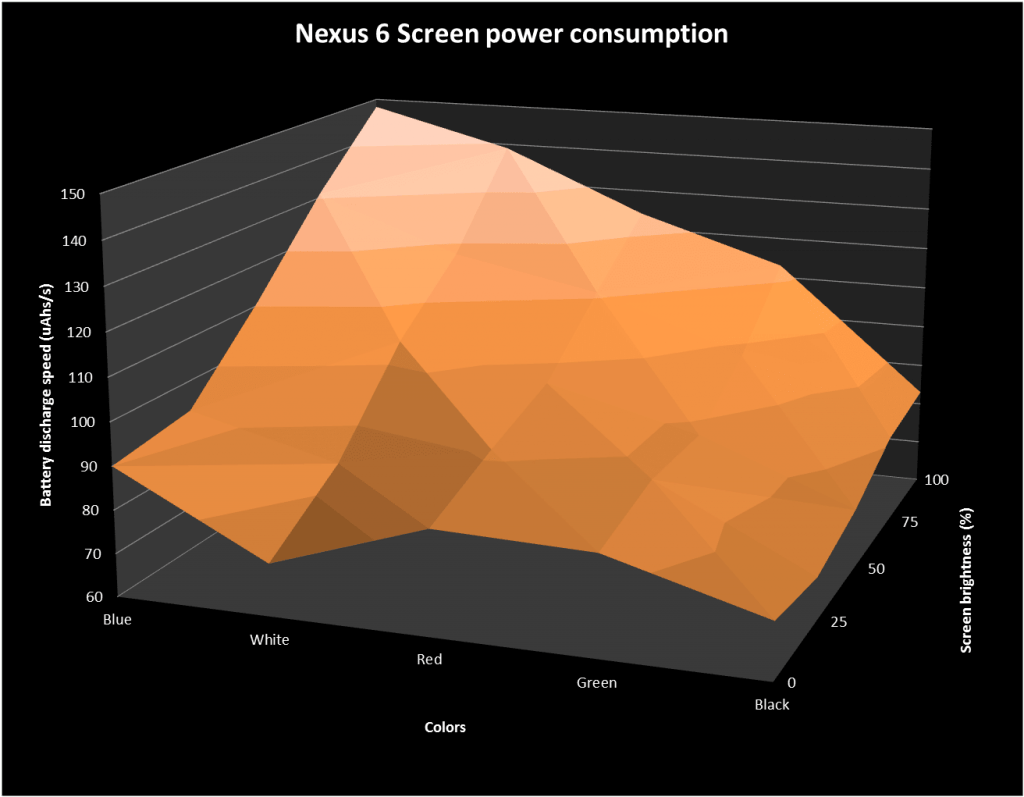
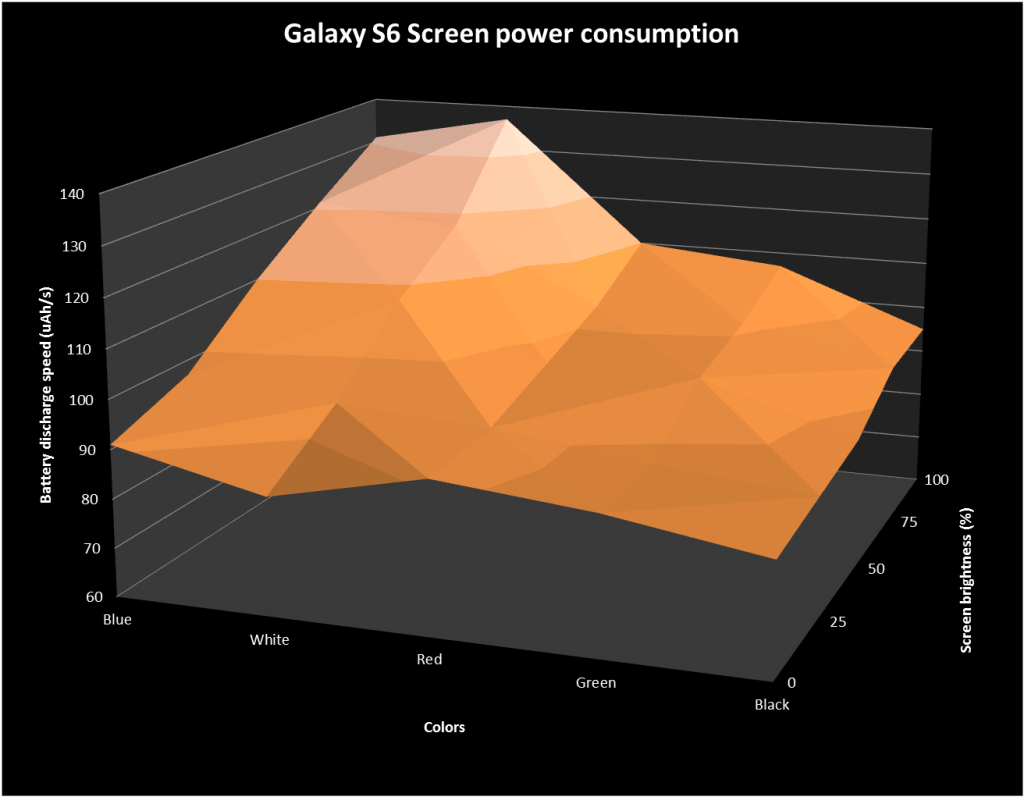
Extreme test shows OLED iPhone X with 'dark mode' saves nearly 60% battery over 3 hours | AppleInsider

Are you rocking the black wallpaper for battery life? Or showing off that awesome OLED screen with something vibrant?! : r/iphone

So I wanted a black wallpaper for my iPhone X but found true black too boring. This is what I found. I think it's by far the cleanest and best looking star

Extreme test shows OLED iPhone X with 'dark mode' saves nearly 60% battery over 3 hours | AppleInsider

I built OLEDify - takes advantage of the iPhone X OLED screen and saves battery by pure-blacking your wallpaper : r/apple